STAKEHOLDER INTERVIEWSPerspectives from 15 global health leaders on GHIT's catalytic role and
Japan's transformational contributions to global health R&D
DEVELOPMENT
03
Dr. Nathalie Strub Wourgaft
Medical Director
Drugs for Neglected Diseases initiative (DNDi)
“For the diseases that have been neglected by research, there are a lot of questions that need answers.”

Why do NTDs matter today?
NTDs are neglected tropical diseases that disproportionately affect the very poor. These diseases have been neglected for decades by pharmaceutical R&D because there is no return on investment to incentivize pharmaceuticals to find solutions for these patients. The term NTD covers a range of infectious diseases that affect patients living in Africa, Latin America, and Asia. These diseases debilitate, disfigure, blind or kill. The poorest of the poor – particularly women and children – are the hardest hit.
The burden of these diseases is significant in terms of morbidity, mortality, lost productivity and impaired economic growth, engendering a vicious cycle of poverty. The World Health Organization (WHO) has established a list of these NTDs to show the need for public commitment to find treatments or vaccines, eliminate the transmission, control and finally eliminate these diseases from the planet.
Why and how was DNDi established?
Founded in 2003 after MSF (Médecins Sans Frontières or Doctors Without Borders) received the Nobel Peace Prize, DNDi is a collaborative, patients’ needs-driven, not-for-profit drug R&D organization.
DNDi aims to develop drugs addressing the needs of patients suffering from the most neglected diseases. It was founded with research institutions from endemic countries where the patients live. MSF was one of the key founders, together with the KEMRI Institute in Kenya, the ICMR (Indian Council of Medical Research) in India, the Malaysian Ministry of Health, Pasteur Institute and the Oswaldo Cruz Foundation/Fiocruz (in Brazil). WHO is a permanent observer of DNDi.
The objective at the time was to limit the scope of the work on kinetoplastid diseases such as sleeping sickness, visceral leishmanaisis (VL) and Chagas disease in order to show that this model of publicprivate partnership could find alternative solutions that the standard pharmaceutical model could not provide because there is no return on investment from the pharmaceutical industry.
As said, DNDi was notably founded by MSF whose doctors had to face diseases in the field for which there were none or no adapted treatments. For instance, for African trypanosomiasis (sleeping sickness) patients were receiving melarsoprol, an arsenic derivative treatment, which killed 1 out of 20 of the patients to which it was given. For a doctor, it is devastating when your only options are interventions that will potentially kill a patient you're trying to save.
Being successful in the delivery of new treatments for sleeping sickness, VL and Chagas disease, DNDi expanded its activities in 2011 to other diseases such as paediatric HIV and filarial diseases. Also, its new Business Plan (2015- 2023) launched a new dynamic portfolio approach with the entry into its R&D portfolio of mycetoma and hepatitis C, two disease areas with significant unmet patient needs.

You mentioned the lack of return on investment for NTD product development. Are there any other challenges or opportunities, scientific of otherwise, in the area of drug development for NTDs?
For the diseases that have been neglected by research, there are a lot of questions that need answers. First, is the question of access to libraries of chemical compounds that could become potential drugs for treatment. One solution is an initiative that was developed by DNDi with the financial support of the GHIT Fund, the NTD Drug Discovery Booster. This has been an incredibly innovative R&D mechanism to expand and enhance research and get access to libraries. The Booster circumvents early stage commercial barriers between the pharmaceutical participants, allowing DNDi, for the first time, to search millions of unique compounds simultaneously, in the hunt for new treatment leads for leishmaniasis and Chagas disease.
You also need to be able to categorize and test whether a potential compound will kill whatever infectious agent you want to kill—parasites, for the most part. So there's a need to build screening projects, which we also did in collaboration with Japanese scientists. Then comes the need for improving the chemistry of the compound so that it can become something you can administer to patients. The next step is to test in the animal models, which will predict human efficacy and safety, where we will also have a big challenge.
Because NTDs have not benefited from a lot of research, we are still trying to find the best models to predict, for example, when you administer a drug in mice that have been infected with a Trypanasoma cruzi parasite (which causes Chagas disease), whatever you see in the mice is predicting something that you will see in humans.
“Infectious disease science existed in Japan, GHIT acted as a catalyzing funding mechanism that allowed this science to emerge and be available for NTDs.”

What other research gaps exist for NTDs?
We have very good models in some areas and much less predictive models in others. Of course, collaboration in building these models is key, because this is how you save time and money—not by investing in clinical trials, but investing more in pre-clinical trials, and then deciding based on preclinical results what you may see in humans. It is very important to have these big models.
When you move to clinical trials, then again there are questions. We think we know how to measure efficacy, but sometimes it takes a very long time, and the more time we spend on clinical development, the more time patients have to wait to access new treatments. We'd like to have clinical trials with earlier markers of efficacy—what we sometimes call surrogate markers—that are again themselves predictive of what you will see in patients. This is a science that has not yet matured very well.
DNDi has already successfully created new drugs for NTDs through cross-sector partnership. In what way have cross-sector partnerships been valuable?
First of all, it’s very clear that no one can do it alone. You need to have partners, but with DNDi we're talking of finding a solution for a shared objective as soon as possible, which requires the flexibility of having the right partner that is doing the right piece of work that needs to be done across a whole spectrum of activities. Each sector brings unique expertise to the table, and our role is to make everyone work together to reach a common goal.
The way we start a project offers a key example. We build a project with what we call a Target Product Profile (TPP). A TPP is defined by members who are part of platforms such as the LEAP Platform for leishmaniasis, the HAT Platform for sleeping sickness or the CCRP Platform for Chagas disease. The platforms bring together all the stakeholders, including those that will be using the product in the field: academics, physicians, national programs, ministries of health, NGOs, and the pharmaceutical industry. Everyone comes together and asks, "Okay, what would be the ideal profile of a drug or treatment for this specific disease? Also, what is acceptable? How far can we go to still accept this as bringing a solution?"
“We hope to show that this funding mechanism results in shared successes, which means that it can be a virtuous cycle, so that maybe other countries learn from the GHIT experience and start building similar processes.”
Please tell us more about DNDi’s relationship with Japan.
DNDi opened an office in Tokyo in 2004 (a year after our creation). In early 2005, we started collaborating with the Kitasato Institute, looking at screening products and natural products from the institute, for targeting products that would kill the trypanosome parasite for sleeping sickness. This was a very exciting and positive collaboration under our first R&D Director, Simon Croft, and it led to the development of a screening tool.
Even before the collaboration with DNDi, Japan had a long history of involvement in infectious diseases. There had been some exchanges between the scientists in the field within Japan and outside of Japan. So that was a natural collaboration.
In what ways has DNDi’s partnership with the GHIT Fund influenced your work and impact—broadly, and in Japan specifically?
The GHIT Fund award has been essential. Infectious disease science existed in Japan, GHIT acted as a catalyzing funding mechanism that allowed this science to emerge and be available for NTDs. Another unique feature of GHIT is that it allows funding of pre-clinical and discovery work, which bears the higher attrition risk and is not the easiest to fund. Looking at the gaps, that is where we need to start, and without it there's no way we'll find any solutions. For this reason, GHIT is really essential and unique in its scope.
Those who are knowledgeable in the field of discovery are highly impressed by the outputs that we have seen already. GHIT was the funding initiator of the NTD Drug Discovery Booster that is unique, efficient, collaborative, and innovative. I am sure we'll see and hear a lot more about this booster.
“What drives me is engaging myself in something that has meaning and has medical value.”
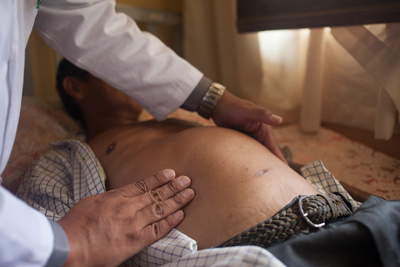
Patient with Chagas disease in Bolivia
In your opinion, how is Japan helping to transform what is possible in vis-à-vis NTD product development?
Japan has a big history in global health. In 2000, it promoted the addition of infectious diseases to the G8 agenda and likewise was very active in promoting the Sustainable Development Goals (SDGs) in the last summit. This past August we were in Nairobi, where a TICAD (Tokyo International Conference on African Development) meeting was organized. It's very clear that Japan has a sustained commitment to global health.
If we look at our own portfolio of collaboration with Japan, there is a lot there. We've had collaboration early on with some research institutes. We also worked with Osaka University, the Tokyo University and Nagasaki University. We've had collaboration in Bangladesh to support a hospital funded by JICA. We have worked with many Japanese companies, including Astellas, Eisai, Takeda, and Shionogi. Additionally, we have collaboration on the immunomodulatorfor cutaneous leishmaniasis, again, with other Japanese companies and universities.
There's a long list of very regular collaborations since we opened our DNDi Japan office in Tokyo in 2004. We've continuously signed new agreements with different actors in Japan. So that in itself, when you look at DNDi's portfolio, it really shows how much Japan has been committed to work with us.
Paint a picture of this collaboration for us, using your partnership with Eisai on E1224 as a lens.
I think the Eisai example shows both the Japanese commitment and the leadership of Eisai's commitment to help the field of NTDs. In 2009, the agreement was signed very quickly to start a study and some work on Chagas disease. E1224 (fosravuconazole, a pro-dug of ravuconazole) is the compound that we've been testing in Chagas disease and also are now testing in Bolivia as a combination treatment.
We also have had some collaboration with Eisai for the NTD Drug Booster, and Eisai was one of the first companies to sign the sharing agreement, which is fantastic. Even more recently, in 2015 and 2016, we have a new collaboration with Eisai on fosravuconzaole to test it for the treatment of Mycetoma. Mycetoma is an absolutely devastating neglected disease. It is fantastic that Eisai has agreed for us to test it in Sudan.

Could you tell us what Chagas disease is, if there are any available drugs, and why new medicine is important?
Chagas disease is an infectious disease that is caused by a parasite that is transmitted by a bug. It is a very nasty bug that lives in the walls and the cracks of the walls of houses in Latin America. It affects millions of people, but most of the time it's a silent disease. After the first infection, most of the patients will not develop an acute disease. Sometimes they will, and they will get a treatment for that, but most of the time they won't. The disease evolves very slowly, and around 20 to 30 years after the first infection some of the patients will develop a cardiac disease, which is essentially untreatable. They will pass away from that disease or they will develop a very worrying digestive-type form of the disease. It's the leading cause of mortality due to infectious diseases in Latin America. So it's a very severe disease.
Some treatments for the disease exist. One is called benznidazole and the other is called nifurtimox. But these treatments, especially for the chronic form, need to be taken twice a day for 60 days, and they are not devoid of side effects. There are major compliance issues because of the long regime and the fact that many patients don’t tolerate them well. What we need is an oral treatment that has higher efficacy and better tolerability. The drug fosravuconazole has a very long half-life and is very unique because after three days of loading dosage it can be given as a weekly treatment.
Now, in fact, to improve the long course of benznidazole, it is combined with fosravuconazole to reduce the duration of treatment, and therefore improve compliance, as a result decreasing the risk of side effects and maintaining or even increasing efficacy in chronic patients.
Looking ahead to the next 5-10 years, what are your hopes for how GHIT and the Japanese pharmaceutical industry, academia, and government will impact NTD drug development?
Sustained GHIT funding is absolutely essential for us. We hope to show that this funding mechanism results in shared successes, which means that it can be a virtuous cycle, so that maybe other countries learn from the GHIT experience and start building similar processes. We would also like to see the GHIT mechanism evolve towards a portfolio funding model that would allow us to seize research opportunities.
What drives your personal engagement in the fight against NTDs?
I’m a medical doctor by training and I’ve been doing drug development for most of my career. What drives me is engaging myself in something that has meaning and has medical value. Doing R&D meets my set of values in doing something that is useful for patients by finding tools to treat them. This is why I’m passionate and fully engaged in medicine and medical research.
The affiliations and positions listed in this interview are at the time of publication of the interview in 2017.

- Biography
- Nathalie Strub Wourgaft, MD, is Medical Director of DNDi, where she has also been Clinical Development Director. Dr Strub Wourgaft has over 15 years of clinical development experience, with Trophos, Pfizer Lundbeck, and the French office of Aspreva, where she held positions of Director of Clinical Development and Medical Director.
STAKEHOLDER INTERVIEWSARCHIVES
FUNDING
-

01
Dr. Naoko YamamotoSenior Assistant Minister for Global Health,
Ministry of Health, Labour and Welfare
#
-

02
Dr. Hannah KettlerSenior Program Officer, Life Science Partnerships
Global Health Program, Office of the President
Bill & Melinda Gates Foundation
#
-

03
Prof. Stephen CaddickDirector, Innovations Division,
Wellcome Trust
#
DISCOVERY
-

01
Dr. David ReddyCEO
Medicines for Malaria Venture (MMV)
#
-

02
Mr. George NakayamaRepresentative Director,
Chairman and CEO
Daiichi Sankyo Company, Limited
#
-

03
Prof. Kiyoshi KitaProfessor Emeritus, The University of Tokyo
Professor and Dean, Nagasaki University School of Tropical Medicine and Global Health
#
DEVELOPMENT
-

01
Mr. Christophe WeberRepresentative Director, President and CEO
Takeda Pharmaceutical Company Limited
#
-

02
Mr. Yoshihiko HatanakaRepresentative Director,
President and CEO
Astellas Pharma Inc.
#
-

03
Dr. Nathalie Strub WourgaftMedical Director
Drugs for Neglected Diseases initiative (DNDi)
#
ACCESS
-

01
Dr. Jayasree K. IyerExecutive Director
Access to Medicine Foundation
#
-

02
Mr. Tetsuo KondoDirector
United Nations Development Programme (UNDP)
Representation Office in Tokyo
#
-

03
Dr. Aya YajimaTechnical Officer
Malaria, other Vectorborne and Parasitic Diseases Unit
Division of Communicable Diseases
World Health Organization Western Pacific Regional Office
#
POLICY
-

01
Dr. Mark DybulFormer Executive Director
The Global Fund to Fight AIDS,
Tuberculosis and Malaria
#
-

02
Dr. Seth BerkleyCEO
Gavi, the Vaccine Alliance
#
-

03
Hon. Prof. Keizo TakemiMember of the House of Councillors of Japan
Chairman, Special Committee on Global Health Strategy
of the Liberal Democratic Party's Policy
#

